The key distinction between solid-state batteries and conventional types lies in their use of solid electrolytes instead of liquid ones. While lithium-ion batteries have advanced significantly, experts agree they've hit performance limits. Solid-state batteries represent the next evolution in battery technology.
Solid-state batteries are smaller, lighter, and deliver higher power density than liquid-electrolyte lithium-ion batteries. The primary hurdles to mass adoption are developing solid electrolytes with adequate ionic conductivity for large-scale batteries and scalable manufacturing processes.
All batteries consist of three core components: the anode, cathode, and electrolyte. They generate power through a chemical reaction where charged ions move from the cathode to the anode via the electrolyte, producing free electrons. This creates a positive charge buildup at the cathode, attracting electrons from the anode to flow through your device, powering it.
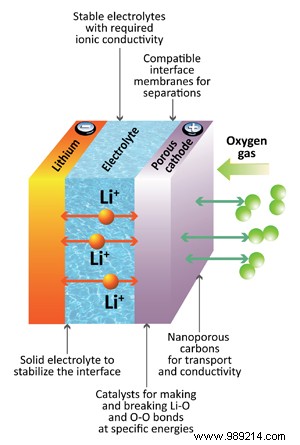
Researchers highlight sodium-based glass electrolytes as a promising lithium-ion alternative, offering three times the energy density. Abundant sodium also minimizes environmental impact compared to scarce lithium materials.
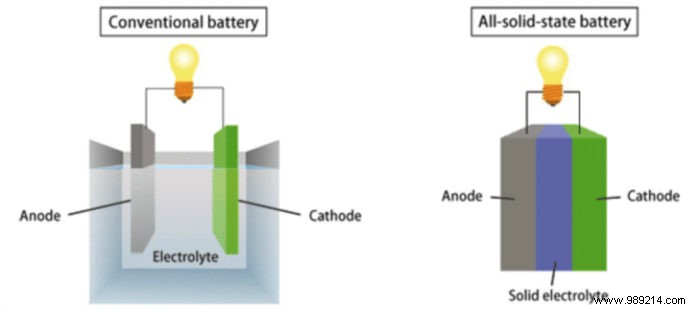
By replacing liquid or polymer electrolytes with solids, these batteries enhance nearly every metric: they're lighter, more eco-friendly, safer (less fire risk), and pack more power. Challenges remain in cost-effective, large-scale production, but ongoing innovations promise economies of scale for widespread use.

Solid-state batteries can upgrade any battery-powered device, but they're game-changers for electric vehicles (EVs). Automakers like Tesla prioritize battery performance, which dictates range and efficiency. Analysts predict EVs will dominate only when solid-state tech matches or exceeds gas-car range, paving the way for mass adoption.
Image credit: Battery with Polymer Separator